
IUPAC’s primary concern is with unequivocal and unambiguous communication. However, in this context, IUPAC does not concern itself with electronic structure. 
This is eminently reasonable, because it is not unexpected that a table based originally on atomic weights does not accord satisfactorily with every nuance of electronic structure. The proponents are often teachers who wish to use a periodic table to facilitate their own teaching of electronic structures. Some of the proposals for the table are baroque in the extreme, but that has not been the direct reason for refusing to adopt them. The Division of Nomenclature and Structure Representation has recently reaffirmed this position. So far, our invariable response has been to refuse, regardless of the merit of the proposal. However, it is really the person using the table who must determine if it meets his or her requirements.Īs an inorganic chemist with experience in nomenclature, I receive many of these new versions, often with a request that IUPAC consider formally approving them. A “left-step” example has recently been mentioned (Scerri, 2008). For these and related reasons, new forms of the periodic table are being continually proposed. 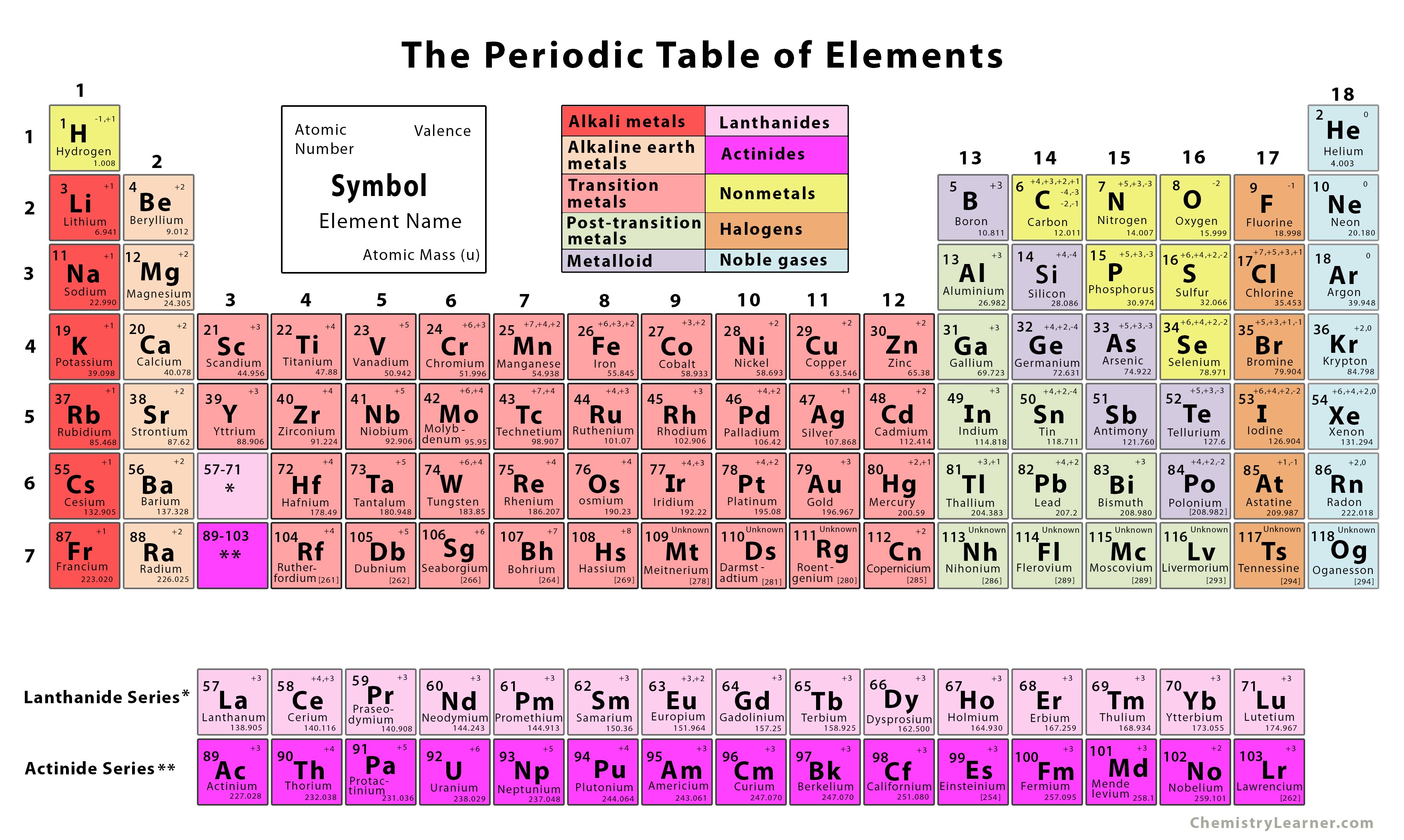
Teachers have sometimes found the long form unsatisfactory for instructing students in detail, and various longer forms are often suggested, for example, to try to regularize the treatment of the lanthanoids and actinoids. None is ultimately the “correct” or “best” shape. 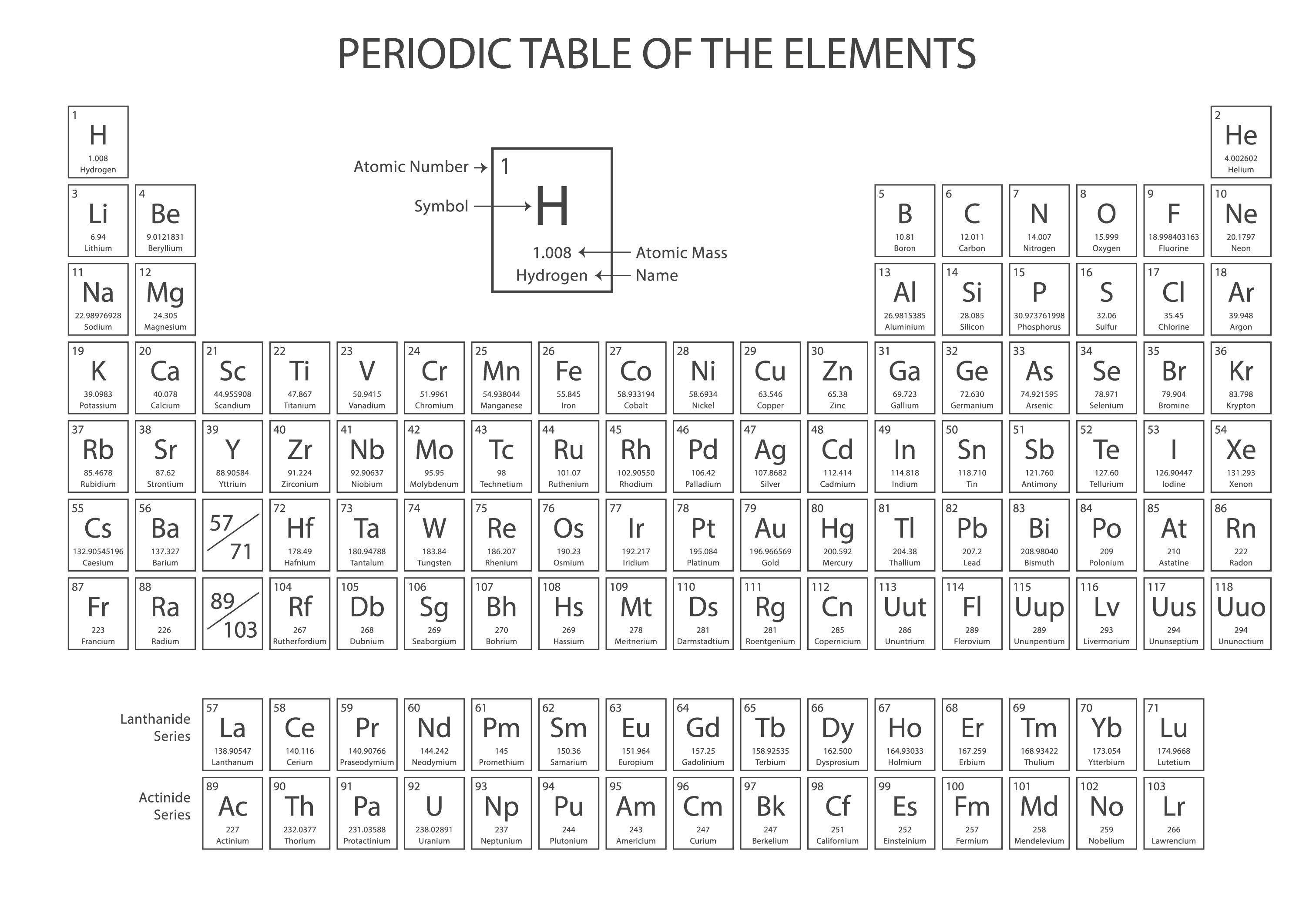
A plethora of shapes have been proposed for the table, both two- and three- dimensional, and triangular or circular or square. Just as the d-transition elements were introduced into the old short form of the table to make a long form (figure 1), the actinoids and lanthanoids have been further inserted to make an even longer form (figure 2). The possibility of producing a “best” form has been discussed recently (Scerri, 2008). Some people have found the simple long-form periodic table unsatisfactory for aesthetic reasons. There is no absolutely satisfactory position for hydrogen (Scerri, 2007). Logically, however, one might consider it as having an outer shell lacking one electron for completion, so that it is also a kind of halogen, maybe a pseudo-halogen, though that term may confuse some readers. In the conventional long form, hydrogen appears as in the first short period along with helium, as a kind of pseudo-alkali metal. As the long form places them, the lanthanoids and actinoids sit rather uncomfortably each in a single place, but should lanthanum and actinium be grouped directly with their congeners? Hydrogen is always a problem. There are minor discrepancies, such as copper seeming to prefer the arrangement d 10s 1, rather than d 9s 2 as one might expect. Once the significance of atomic electronic configurations was realized, the table was adapted to be almost completely consistent with them. The periodic table was developed from considerations of chemical properties and atomic weights and even then the latter arrangement was not without inconsistencies. For example, it is not strictly consistent with all our ideas about electronic structure. Teachers especially find that it does not fulfill all their requirements. Nevertheless the conventional long form with the 1–18 group numbering, which goes back at least to the 1920s (Paneth, 1923), is certainly not ideal for every purpose. The usual presentation of the periodic table which we use (figure 1) is subject to continual updating as new elements are produced (Holden and Coplen, 2004). The implication is that this is an IUPAC-approved table (see, for example, Lavelle, 2008), and indeed a Google search of the Web will unearth several entries for “IUPAC approved Periodic Table.” Figure 1 is based upon the periodic table that appears on the IUPAC website, though nowhere is it stated that that version is “approved.” In fact, IUPAC has not approved any specific form of the periodic table, and an IUPAC-approved form does not exist, though even members of IUPAC themselves have published diagrams titled “IUPAC Periodic Table of the Elements.” However, the only specific recommendation IUPAC has made concerning the periodic table covers the Group numbering of 1–18. The so-called short form cannot display the 1-18 numbering, and has largely fallen into disuse. Invariably this version of the table is what was once known as the long form, in which the various Groups are numbered from 1 to 18 (figure 1). In more than one IUPAC publication you will find a periodic table, sometimes referred to as an IUPAC Periodic Table. A table showing the periodicity of the properties of many chemical elements, from the first English edition of Dmitrii Mendeleev’s Principles of Chemistry (1891, translated from the Russian fifth edition).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
